Investigating the possibilities of bacteriophages: How these viruses may aid in combating antibiotic resistance
In a world where the threat of antibiotic-resistant bacteria looms large, a growing number of scientists are turning to a surprising ally in the fight against superbugs—viruses. But not the kind that cause illness in humans. These are bacteriophages, or simply “phages,” viruses that specifically infect and destroy bacteria. Once sidelined by the success of antibiotics, phage therapy is now being re-evaluated as a promising alternative as the medical community grapples with drug resistance.
The notion of employing viruses to combat bacterial infections might appear unusual, yet it is based on scientific principles established more than 100 years ago. Phages were initially identified by British bacteriologist Frederick Twort and French-Canadian microbiologist Félix d’Hérelle in the early 1900s. Although the concept gained traction in certain areas of Eastern Europe and the ex-Soviet Union, the introduction of antibiotics in the 1940s caused phage research to decline in prominence within Western medical practices.
Ahora, con la resistencia a los antibióticos transformándose en una crisis de salud mundial, el interés en los fagos está resurgiendo. Cada año, más de un millón de personas en todo el mundo fallecen a causa de infecciones que ya no responden a los tratamientos habituales. Si esta tendencia persiste, esa cifra podría ascender a 10 millones al año para 2050, poniendo en riesgo muchos aspectos del cuidado médico moderno, desde cirugías comunes hasta terapias contra el cáncer.
Phages provide a distinct answer. In contrast to broad-spectrum antibiotics, which eliminate both harmful and beneficial bacteria without distinction, phages exhibit high specificity. They attack particular bacterial strains, leaving nearby microorganisms unaffected. This accuracy not only minimizes unintended harm to the body’s microbiome but also aids in maintaining the long-term efficacy of treatments.
One of the most exciting aspects of phage therapy is its adaptability. Phages reproduce inside the bacteria they infect, multiplying as they destroy their hosts. This means they can continue to work and evolve as they spread through an infection. They can be administered in various forms—applied directly to wounds, inhaled to treat respiratory infections, or even used to target urinary tract infections.
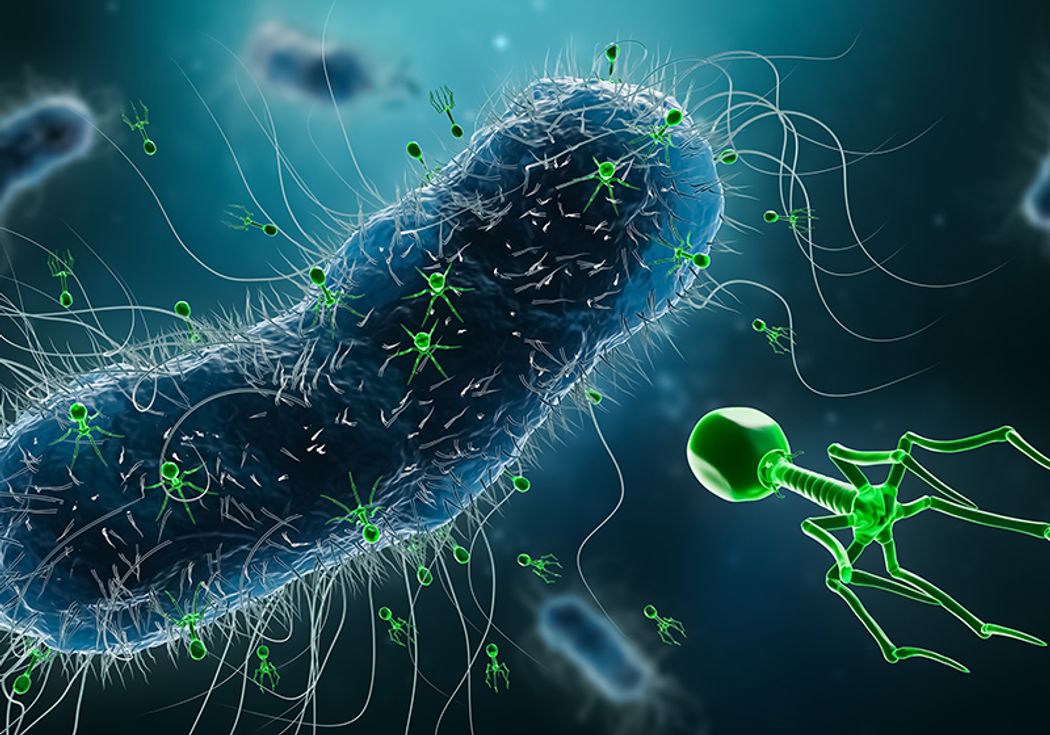
Research laboratories worldwide are investigating the healing possibilities of phages, and a few are welcoming public involvement. Researchers at the University of Southampton participating in the Phage Collection Project aim to discover new strains by gathering samples from common surroundings. Their goal is to locate naturally existing phages that can fight against tough bacterial infections.
The procedure for identifying useful phages is both unexpectedly simple and scientifically meticulous. Participants gather samples from locations such as ponds, compost piles, and even unflushed toilets—any spot where bacteria prosper. These samples are filtered, processed, and then tested with bacterial cultures from actual patients. If a phage in the collection destroys the bacteria, it might be considered for future treatment.
What makes this approach so promising is its specificity. For example, a phage found in a home environment might be capable of eliminating a strain of bacteria that is resistant to multiple antibiotics. Scientists analyze these interactions using advanced techniques such as electron microscopy, which helps them visualize the phages and understand their structure.
Phages look almost alien under a microscope. Their structure resembles a lunar lander: a head filled with genetic material, spindly legs for attachment, and a tail used to inject their DNA into a bacterial cell. Once inside, the phage hijacks the bacteria’s machinery to replicate itself, ultimately destroying the host in the process.
However, the path from identifying to treating is intricate. Every phage has to be paired with a distinct bacterial strain, a process that requires time and experimentation. In contrast to antibiotics, which are produced on a large scale and have wide-ranging applications, phage therapy is usually customized for each patient, complicating the regulatory and approval processes.
Despite these challenges, regulatory bodies are beginning to support the development of phage-based treatments. In the UK, phage therapy is now permitted on compassionate grounds for patients who have exhausted conventional options. The Medicines and Healthcare products Regulatory Agency has also released formal guidelines for phage development, signaling a shift toward greater acceptance.
Experts in the field stress the importance of continued investment in phage research. Dr. Franklin Nobrega and Prof. Paul Elkington from the University of Southampton emphasize that phage therapy could provide vital support in the face of increasing antibiotic resistance. They highlight cases where patients have been left with no effective treatments, underscoring the urgency of finding viable alternatives.
Clinical trials are still needed to fully validate phage therapy’s safety and efficacy, but there is growing optimism. Early results are encouraging, with some experimental treatments showing success in clearing infections that had previously defied all conventional antibiotics.
Beyond its possible applications in medicine, phage therapy introduces a fresh approach to involving the public in scientific endeavors. Initiatives such as the Phage Collection Project encourage individuals to participate in scientific research by gathering environmental samples, fostering a sense of participation in addressing one of the critical issues of our era.
This local effort may be crucial in discovering novel phages that could be vital for upcoming therapies. As the globe deals with the escalating challenge of antibiotic resistance, these tiny viruses might turn out to be unexpected saviors—evolving from little-known biological phenomena into critical instruments of contemporary medicine.
Looking to the future, there is optimism that phage therapy might become a regular component of medical treatments. Infections that currently present significant threats could potentially be addressed with specifically tailored phages, delivered efficiently and securely, avoiding the unintended effects linked with conventional antibiotics.
The journey ahead will necessitate collaborative actions in the realms of research, regulation, and public health. However, armed with the tools of molecular biology and the zeal of the scientific community, the promise of phage therapy to transform infection management is tangible. What was once a disregarded scientific notion may shortly become central in the fight against antibiotic-resistant diseases.